Article Text
Abstract
Background Training health providers is an important strategy to improve health. We conducted a cluster-randomised two-arm trial in Tanzania to assess the effect of a 1-day competency-based training ‘Helping Mothers Survive Bleeding after Birth (HMS BAB)’ followed by eight weekly drills on postpartum haemorrhage (PPH)-related morbidity and mortality.
Methods Twenty districts in four purposefully selected regions in Tanzania included 61 facilities. The districts were randomly allocated using matched pairs to ensure similarity in terms of district health services in intervention and comparison districts. In the 10 intervention districts 331 health providers received the HMS BAB training. The other half continued with standard practices. We used the WHO’s near miss tool to collect information on severe morbidity (near misses) of all women admitted to study facilities. We performed interrupted time series analysis to estimate differences in the change of near miss per delivery rate and case fatality rates. We also assessed implementation of evidence-based preventive and basic management practices for PPH as secondary outcomes.
Results We included 120 533 facility deliveries, 6503 near misses and 202 maternal deaths in study districts during study period (November 2014 to January 2017). A significant reduction of PPH near misses was found among women who suffered PPH in the intervention district compared with comparison districts (difference-in-differences of slopes −5.3, 95% CI −7.8 to −2.7, p<0.001) from a baseline PPH-related near miss rate of 71% (95% CI 60% to 80%). There was a significant decrease in the long-term PPH near miss case fatality (difference-in-differences of slopes −4 to 0) (95% CI −6.5 to −1.5, p<0.01) in intervention compared with the comparison districts. The intervention had a positive effect on the proportion of PPH cases treated with intravenous oxytocin (difference-in-differences of slopes 5.2, 95% CI 1.4 to 8.9) (p <0.01).
Conclusion The positive effect of the training intervention on PPH morbidity and case fatality suggests that the training addresses important deficits in knowledge and skills.
Trial registration number PACTR201604001582128.
- near miss
- postpartum haemorrhage
- PPH near miss
- competency-based training
- Tanzania
- maternal morbidities and mortalities
This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, appropriate credit is given, any changes made indicated, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/.
Statistics from Altmetric.com
- near miss
- postpartum haemorrhage
- PPH near miss
- competency-based training
- Tanzania
- maternal morbidities and mortalities
Key questions
What is already known?
The Helping Mothers Survive Bleeding after Birth (HMS BAB) training is implemented in varying degrees in a number of low/middle-income countries and studies suggest an improvement in knowledge, skills and confidence to manage postpartum haemorrhage (PPH).
Evidence is missing on whether the training results in improvements in maternal health outcomes (near misses and mortality).
What are the new findings?
We report small but significant improvements in immediate and long-term reduction of PPH near misses and PPH case fatality rates in intervention compared with comparison districts suggesting that the training makes an important impact on morbidity and mortality reductions.
Overall case fatality rates were still high as access to advanced life-saving interventions is very limited.
What do the new findings imply?
The HMS BAB training, although effective, requires additional support to ensure necessary drugs, equipment and supplies are available to bring sustained positive outcomes.
Background
Despite efforts to address maternal health challenges, most countries with a high burden of poor maternal health outcomes did not meet Millennium Development Goal 5 targets by 2015—Tanzania is no exception. Even with renewed efforts to reach targets set in the Sustainable Development Goals by 2030, maternal mortality remains a global challenge, especially in low/middle-income countries. In 2015 alone, approximately 300 000 women died of pregnancy and childbearing-related causes, a quarter of which were caused by postpartum haemorrhage (PPH).1 In Tanzania, maternal mortality is estimated at 398 deaths per 100 000 live births with 25% of all maternal deaths due to haemorrhage.1 2
Giving birth in a facility, attended by a skilled health provider and within a functioning health system have been identified as critical components for reducing high mortality rates.3 There has been a corresponding increase in facility deliveries worldwide.4 However, a few country analyses have indicated that despite increases in facility births, reductions in mortality rates have remained insufficient.4–6 In response, there is an increased focus on improving the quality of care in facilities, especially in low/middle-income countries7–10 and to address the low competency levels among health providers in obstetrics including preventing and managing PPH.11–15
In-service training has long been used to improve health workers’ competences with varying degrees of success. More recent advances in educational research have led to the conceptualisation of competency-based training supported by simulations to make learning more effective.16 17 One such training is a 1-day competency-based Helping Mothers Survive Bleeding after Birth (HMS BAB) programme using a low-fidelity simulator—the Mama Natalie.18
Evidence suggests that the HMS BAB training conducted in low/middle-income countries improves providers’ knowledge and skills competency on PPH prevention.13 19–21 While the training has been taken to scale in several countries21 22 evidence is missing on whether the training does in fact improve maternal outcomes. To provide data on the effect of the training on maternal outcomes, we conducted two trials in the two countries of Uganda and Tanzania. Here we report on the cluster-randomised trial evaluating the effect of the HMS BAB training on morbidity and case fatality rates in Tanzania.
Methods
Study design
This cluster-randomised trial including 20 clusters (districts) was conducted to assess the effect of the HMS BAB training on changes in rates of severe maternal morbidity—defined as maternal near miss, among mothers experiencing PPH in Tanzania. Clusters (districts) were used for the level of randomisation because trainings bring effects through prevention, management and referral, beyond the individual care for women.23 The HMS BAB training was provided to 31 facilities in 10 intervention clusters while comparison clusters continued with standard care. A cluster was an administrative health district. Details are presented in the trial protocol paper.23
Study setting
The study was done in four regions of Tanzania: Simiyu and Mwanza (Lake zone) and Lindi and Mtwara (Southern zone). The regions were purposefully selected by the Ministry of Health, Community Development, Gender, Elderly and Children. The proportion of women delivering in a health facility according to the latest Demographic and Household Survey was 32% (Simiyu), 53% (Mwanza), 80% (Lindi) and 81% (Mtwara).24 Basic emergency obstetric and neonatal care signal functions are not consistently implemented in hospitals and health centres throughout the country, but all hospitals and selected health centres do provide caesarean section services.25–27 Eighty-eight per cent of facility births are conducted by various nurse and nurse-midwife cadres or medical assistants and 12% by clinicians (clinical officers, assistant medical officers and medical officers).
Study sites and participants
Inclusion criteria for a district (cluster) were having a district hospital and two to three large health centres where at least 3000 deliveries are attended per year, and no other scheduled capacity building for obstetric care. Overall, we included 20 districts with 61 health facilities. We included all women attending the selected facilities for delivery care or obstetric complications from July 2015 to November 2016. All providers working in selected maternity wards and present during the training were enrolled as secondary participants in the trial.
Intervention
The intervention, the HMS BAB training, was developed by Jhpiego and Laerdal Global Health and includes prevention and basic care for PPH. The basic training which we evaluated has recently been complemented by an advanced training module.28 The curriculum of the basic HMS BAB training is described in Box 1. The content is aligned to what is taught in preservice trainings, and aims to reinforce and refresh skills rather than introduce new knowledge and skills. For each facility, training consisted of an initial 1-day training, followed by weekly drill sessions for 6–8 weeks. These short but frequent training sessions aim to build on learnt skills and enhance skills competency. These ‘low dose, high frequency’ learning sessions are facilitated by a pair of peer practice coordinators in each facility. Facilities in comparison districts continued with standard practice and received the HMS BAB training at the end of the trial (November 2016).
Helping Mothers Survive Bleeding after Birth (HMS BAB) competency-based training
The HMS BAB intervention
A competency-based 1-day training using a low-fidelity birthing simulator: Mama Natalie, https://www.laerdal.com/products/simulation-training/obstetrics-paediatrics/mamanatalie/
The curriculum includes basic delivery care and active management of the third stage of labour (AMTSL), a step-by-step assessment of the mother’s condition and blood loss and basic management of postpartum haemorrhage (PPH) including application of intravenous oxytocin, uterine massage and recognition of when to refer and referral preparedness.
In-facility (different from centrally organised training where a few providers attend from each facility) in-service training where all maternity providers within the facility are trained.
Uses ‘low dose, high frequency’ teaching methodology where the initial 1-day training is supplemented by eight weekly sessions to practise scenario-based drills that build on learnt competencies. These sessions are supported by a pair of facility-based peer practice coordinators.
Our theory of change is depicted in figure 1. We postulate that the training intervention and the frequent drills will improve providers’ knowledge and skills. The training should also improve emergency preparedness and the preparation of guidelines for referral at facilities. Improved knowledge and skills and emergency preparedness should lead to consistent implemented preventive practices of active management of the third stage of labour (AMTSL), enhanced recognition of PPH and early intervention for PPH such as application of oxytocin. The drills should also stimulate a better learning environment within the facility even when new providers are posted and ultimately will have a sustained effect on skills. Hence, facilities receiving the HMS BAB training will have reduced PPH case fatality and PPH near misses.
Theory of cChange. AMTSL, active management of the third stage of labour; HMS BAB, Helping Mothers Survive Bleeding after Birth; PPH, postpartum haemorrhage.
Implementation
The implementation followed a stepwise approach with three main steps (figure 2). Step 1 where Jhpiego-trained ‘master trainers’ facilitated the training of 12 district trainers in a central training. Step 2, the district trainers were accompanied by Jhpiego master trainers to intervention districts. The district trainers were paired, each pair conducted their first in-facility training under the supervision of Jhpiego master trainers to ensure their adherence to training standards. Each pair spent 1.5 days per facility conducting the training. Day 1 was for the HMS training to all providers and day 2 was spent in coaching two local providers to become facility peer practice coordinators. Providers who excelled in the skills training, were willing to lead others and had good communication skills were selected to be peer practice coordinators and received this half-day training and coaching. After this, they were expected to facilitate the six to eight weekly drills using specific scenarios to augment the learnt skills (step 3). A drill session is expected to last approximately 30–40 min.
Timeline of the trial. HMS BAB, Helping Mothers Survive Bleeding after Birth.
The in-facility training (step 2) was done between 15 January and 3 February 2015 and 331 providers in 31 health facilities in the 10 randomly selected districts were trained. Facility and labour ward administrators were informed about the date of training a week in advance. Providers who were off duty or scheduled to work different shifts on those dates were still requested to attend the training. When available, district reproductive health coordinators or their representatives accompanied trainers to ensure the involvement of district administrators for mentoring capacity. The district trainers made an additional supervision visit and weekly calls to peer practice coordinators to follow-up during the first 3 months after step 2 training.
All trainings used the standard HMS BAB curriculum and training materials; a set of the materials including a Mama Natalie was donated to each of the facilities.
Outcomes
We defined three primary outcome indicators: (1) all-cause near misses among all women who delivered in the facility, (2) PPH near misses among all women who delivered in the facility, and (3) PPH near misses among women who suffered PPH during facility delivery. We assessed changes in the trends of the occurrence between intervention and comparison district cluster-level estimates.23 We defined PPH near miss case fatality as a death of a mother who experienced PPH near miss. We defined secondary outcome indicators as (1) interventions provided to prevent and manage PPH such as: AMTSL, oxytocin administration, manual removal of placenta, receipt of a blood transfusion, hysterectomy, and (2) changes in providers’ knowledge and competencies in PPH care. The latter was assessed using a standard knowledge questionnaire as well as a checklist of observations from the simulated drills. Thus, we included a comprehensive evaluation from reaction to effects along the implementation pathway according to the Kirkpatrick model.29
Sample size
We assumed all-cause near miss and PPH near miss prevalence of 6 and 2 per 100 deliveries, respectively30 31 and aimed to observe a 25% reduction in the proportion of the baseline primary indicator at 80% power and 5% error and an intercluster factor (k) of 0.15. The sample size was 10 clusters in each trial arm, with 3000 deliveries per cluster.23
Randomisation
Districts were enrolled after consultation with regional administrations and on fulfilment of inclusion criteria.14 We matched the districts using information from a baseline health facility survey and baseline near miss data collection which included: number of hospitals and health centres per district, rural/urban districts, Lake and Southern zone districts and proportion of primary outcomes reported during the baseline period. Ten matched district pairs were created, half of each pair was randomly allocated to be in either the intervention or comparison arm of the trial. A biostatistician, independent of the implementation team, randomised and allocated clusters to reduce selection bias (used STATA software V.15). The intervention did not allow for blinding of implementers or providers.
Data collection
We collected near misses from all enrolled facilities prospectively. We adapted the WHO’s disease and management-based near miss tool for data collection and included background information on age, parity, induction/augmentation of labour and estimated blood loss.31 32 We estimated the amount of PPH blood loss using visual observation and encouraged providers to employ the kanga (traditional cloth) method which uses a number of soaked kangas to estimate blood loss.33
We trained two nurse-midwives or clinicians as data collectors per facility. Training, retraining and supervision of data collectors are described in the protocol paper.23Abstracted data were entered electronically (Lenovo tablets A3500F) and uploaded to a secure server on a weekly basis. Continuous data checks on form completion and upload status were done by the national study coordinator.14 The national study coordinator made monthly telephone calls to collect information on all facility deliveries. The coordinator also made supervision visits at first every month, then every quarter to verify the data collected against the facility registers, including looking at case notes. All data collectors were connected to a WhatsApp message group for continuous support.
We conducted two health facility assessments, one at baseline (May 2015) and one postintervention (October 2016), to understand the district health infrastructure and readiness for PPH care. We used a modified checklist from Tanzania service provision assessment tools combined with information from similar in-country studies to assess facility readiness to provide PPH interventions.14
In addition, all providers who attended the training completed a before-and-after assessment on knowledge and competencies on detecting and managing PPH.19 20
Statistical analysis
We used STATA software V.15 for all analysis and based the analysis on ‘intention to treat’. The unit of the analysis was the district. Descriptive variables obtained from individual health facility assessments were collapsed to reflect district estimates. Numerical variables were presented as medians with IQRs, and categorical variables were presented as proportions with 95% CIs. Mann-Whitney non-parametric test and χ2 test were used to compare the two trial groups at baseline and postintervention periods. P values less than 0.05 were considered significant.
For the effect evaluation we included 6-month baseline and 10-month postintervention assessment in all 20 districts. Interrupted time series analysis was used to estimate seven parameters for each primary and secondary outcome.34
The preintervention trend (slope) in the comparison districts.
The difference in the mean (intercept) outcome between intervention and comparison districts prior to the intervention.
The difference in the slope (trend) of the outcome between intervention and comparison districts prior to the intervention.
The change in the level of the outcome that occurs in the period immediately following the introduction of the intervention in the comparison districts (short-term effect).
The difference between preintervention and postintervention slopes of the outcome (long-term effect) in the comparison districts.
The difference between intervention and comparison districts in the level of the outcome variable immediately following introduction of the intervention (short-term effect).
The difference between intervention and comparison districts in the slope (trend) of the outcome after initiation of the intervention compared with preintervention (long-term effect).
We used interrupted time series analysis in order to compare the trends over time in the outcomes of interest, between intervention and control clusters before and after the intervention period and, by doing so, to adjust for baseline differences between intervention and comparison groups.
Ethical considerations
The study received ethical clearance from the Senate Research and Publication Committee of Muhimbili University of Health and Allied Health Science. Written informed consent to include all facilities was gained from the district medical officers as well as the medical officers in charge of the selected facilities. Written consent was gained from all providers who were trained and were assessed during the intervention. No consent was sought from individual women as we only used facility data routinely collected in case notes and registries. All data were deidentified prior to electronic entry and storage.
Results
The trial took place in all of the intervention and comparison facilities enrolled, with no facility dropping out. Two of the 200 postintervention months of data collection were missed as facilities failed to report data (one each from an intervention and comparison district); both were remote low case load facilities. We imputed the missing values by calculating the average of district-month data points lying before and after the missing data point. All facilities, except for two, were trained in January 2015. Two were delayed due to one facility being unreachable due to rain, and the other experiencing an acute staff shortage due to competing official work. Training was held in these two facilities in February 2015.
Table 1 shows that intervention and comparison districts had similar levels of facility readiness at baseline and postintervention surveys. Most labour wards had oxytocin available: 89% (95% CI 89% to 100%) in intervention district and 89% (95% CI 77% to 100%) in comparison district, respectively. Referral readiness was insufficient in both, with less than half of the facilities having functional transport on the day of the assessment.
Facility readiness to care for women with PPH: staffing, case load and infrastructure by intervention and comparison districts
We trained 331 providers of different cadres working in the maternity sections: 12% were clinicians, 67% nurse-midwives, 19% medical attendants and six laboratory technicians and nursing students. Eighty per cent completed the training (268/331). Table 2 shows the mean pretraining PPH knowledge score of 74% which increased by 13% points after the training (95% CI 10 to 16). Skills assessment (from simulations) showed the following gains: 49% point increase for AMTSL (95% CI 41 to 57), 42% point increase for recognition of retained placenta (95% CI 32 to 50) and 42% point increase (95% CI 39 to 45) for management of severe PPH.
Knowledge and competencies (from simulations) before and after HMS BAB
We included 120 533 facility deliveries in our data set (66 380 in intervention and 54 173 in comparison districts). Facilities reported 6503 cases of all-cause near misses and 202 maternal deaths during the trial period (figure 3). We saw no clinically relevant differences in relation to age and parity of women included in the study (table 3).
Sociodemographic characteristics of women who were included into the study
Trial flow chart. PPH Postpartum hemorrhage
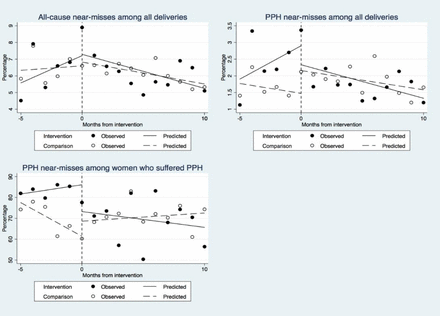
We observed no significant differences in two of the primary indicators: all-cause near misses and PPH near misses among all women who delivered in a facility (table 4, figure 4). There was a significant downward trend of PPH near miss cases among women who suffered PPH in facility delivery in intervention districts compared with the comparison districts (difference-in-differences of slopes −5.3, 95% CI −7.8 to −2.7, p<0.001). Among secondary indicators, the interrupted time series analysis indicated that more women who suffered PPH were treated with intravenous oxytocin (difference-in-differences of slopes 5.2, 95% CI 1.4 to 8.9) (p<0.01) and fewer women received a blood transfusion (difference-in-differences of slopes −8.0, 95% CI −12.6 to 3.4, p<0.01) in the intervention compared with comparison districts (table 4, figure 5). Differences in the intercept and short-term and long-term trends are shown in the online supplementary table. We observed no difference between intervention and comparison districts for other secondary indicators which included: discharge with haemoglobin<70 g/L, removal of retained placenta or women treated with hysterectomy.
Supplemental material
Change of trends in the three primary outcome indicators from baseline to postintervention. PPH, postpartum haemorrhage.
Change of trends of the secondary outcome indicators from baseline to postintervention. AMTSL, active management of the third stage of labour; BT, blood transfusion; PPH, postpartum haemorrhage.
Effects of the HMS BAB training on primary and secondary indicators using interrupted time series analysis
A total of 81.5% of women who suffered PPH during facility delivery were reported to have had received AMTSL and 92.4% received intravenous oxytocin during baseline data assessment (comparison group).
A similar table showing differences in indicators at baseline, short-term and long-term effects between intervention and comparison districts is shown in the online supplementary table.
We reanalysed data using a modified near miss definition to include cases with two units of blood transfused (compared with one unit) as advised by the data advisory board. Our results were weakened and lost significance though the direction of change remained constant (online supplementary figure 1).
Supplemental material
Discussion
Main findings
We observed a strong protective short-term and long-term effects of the training on PPH near misses among women who suffered PPH during health facility delivery in intervention compared with comparison districts. Importantly, we also saw significant reductions in case fatality in PPH cases in the intervention compared with the comparison districts, although a small effect. There was no evidence of any effect on all-cause near misses, PPH near misses in all deliveries or occurrence of PPH in all deliveries.
Our study was conducted in typical rural districts in two distinct zones in Tanzania, which makes the study findings applicable to other areas of Tanzania. Our findings imply that once a PPH occurred, a lower proportion of women deteriorated enough to be classified as PPH near misses. We hypothesise, taking our theory of change into consideration, first, that recognition of severe bleeding improved as the training emphasised blood loss estimation, enhanced early PPH detection and increased vigilance and diagnosis of cases. This was also reported in other competence-based training studies.35–38 Second, our post-training assessment indicated that health providers were more competent to diagnose and manage PPH following the frequent practised drills. This has also been shown by other studies.19 21 22 35 39 As expected, once PPH was diagnosed, health providers could intervene early and prevent women deteriorating to a more severe condition. Third, the training included all providers working in the maternity sections. This inclusive, holistic approach of the training may have promoted continued learning and teamwork within the facility which further enforced the first two effects.
We observed high implementation levels for AMTSL as well as management of PPH with oxytocin, which is encouraging. While we cannot exclude the possibility of a reporting bias (as our estimates are based on health providers’ reports), estimates compare well with those from an observational study in Tanzania that also reported proportions of AMTSL implementation above 90% .35 Our pretraining assessment indicated a reasonable baseline knowledge of prevention and treatment of PPH while skills scores were lower. Various emergency obstetric care training courses have been implemented in the country and health workers are aware that AMTSL ought to be done.13 36 40 The reported high implementation levels of AMTSL could also be the reason that we could not see a reduction of PPH in this trial as other studies evaluating HMS BAB did.35
Similar to this trial, a different simulated training in Mexico, Programa de Rescate Obstétrico y Neonatal Tratamiento Óptimo y Oportuno, reported improved clinical practices but no reduction in obstetric haemorrhage cases41—probably also because AMTSL was already well implemented.
We found a significant reduction in the proportion of women receiving a blood transfusion following PPH in intervention districts, similar to those reported elsewhere.37 We observed an improved availability of blood in both trial groups during the trial period, following a change in blood collection and distribution services from zonal to district level in Tanzania. This supports the hypothesis that early recognition and administration of intravenous oxytocin may have reduced the severity of the PPH. However, it is important to note that most women only received a single unit of blood transfusion (median 1.5, IQR 1–2, data not shown) as there is large demand on blood supplies for other conditions such as pregnant women with anaemia (11% of reproductive aged women have moderate anaemia and 1%–2% have severe anaemia)24 as well as paediatric conditions or HIV/AIDS. The very restricted use of blood transfusion is also supported by our finding that almost 20% of women who had their haemoglobin checked were discharged with a level below 70 g/L and thus with a suboptimal treatment.
While we saw an encouraging increase in the use of prompt basic management intervention, we observed very few cases where additional life-saving interventions such as intrauterine balloon tamponade, B-lynch sutures, drugs like misoprostol or tranexamic acid were used. More advanced management and technology needs to be implemented, however, consistently to reduce morbidity and mortality further. Since the implementation of this HMS BAB training a second module was developed and tested which includes additional elements such as manual removal of the placenta, cervical laceration repair and advanced medical management including use of tranexamic acid (the module is called ‘HMS BAB complete’).28
Strength and limitations
The strength of this trial is that we included 61 facilities and over 120 000 deliveries from both the northern and southern parts of the country. While we aimed to include ‘typical’ districts, the structure however differed slightly with some districts having more hospitals or facilities offering caesarean section. This created imbalances within the trial population including the total number of deliveries. However, we adjusted in the interrupted time series analysis for these baseline imbalances. We present our trial as an effectiveness trial which was undertaken in typical conditions with resource constraints. Our two facility assessments indicated severe shortages in drugs, supplies and the referral system; there was no evidence of difference in the overall resource level between the two trial arms. Three health centres were upgraded to become comprehensive emergency obstetric care facilities and one health centre was upgraded into a district hospital as part of the national road map to improve maternal and newborn health. Consequently, the number of deliveries in these facilities increased. We also report on improvements: availability of blood and the number of facilities equipped to provide emergency obstetric care increased during the trial, including having a written referral plan. These concurrent improvements were part of the nationwide efforts to improve maternal and newborn health and might have biased our results, probably towards weakened estimates of effect.
Our evaluation was based on the theory of change proposed by Kirkpatrick. We report on (1) improved knowledge and skills (increased simulated skills scores, table 2) and increased availability of protocols (table 1); (2) improved application of curative interventions (intravenous oxytocin and reduced need for blood transfusion, table 4); and (3) reduced PPH near miss rate, reduced case fatality rate and thus mortality and morbidity outcomes (table 4).29 This is different from most other evaluations which only reported a positive effect on knowledge and skills but were not designed to indicate improved practices and any effects on mortality and morbidity outcomes.13
We used an accepted methodology to define morbidity using the WHO near miss definition. Our near miss estimate of 6.8 near misses per 100 deliveries compares well with other estimates.31 32 42–44 Tunçalp and Souza in a systematic review reported a prevalence of 0.6%–14.9% using disease-specific criteria and 3.15% using a combination of disease and management-specific criteria in African countries.45 We included as near misses all those cases with one or more blood transfusions as a management criterion as proposed by Nelissen et al (2013)31 and reanalysed using two blood transfusions. We believe that any blood transfusion is a marker of severity in districts reporting shortage of supply of blood.
We relied on trained maternity staff to collect data which is in contrast to most other near miss studies who employed research nurses. This might have affected the completeness and reliability of the data, and could have resulted in reporting bias. Two other studies in the region which assessed maternal near miss prevalence and quality of care have used health facility staff involved in maternity care for data collection with study staff supervision, and reported acceptable levels of data completion and accuracy.46 47 We performed regular supervision visits to verify the reported data against registers and case notes. Also, regular mobile calls by our study team aimed to support the completeness and accuracy of reporting. In addition, a quality assurance exercise carried out in a third of the facilities showed only small differences between captured cases and registers/case notes. We report on documented practices and outcomes, thus relying on appropriately filled registers and case notes which is itself a challenge due to potential under-reporting as health workers prioritise patient care above documentation if the case load is high. However, as our estimates of PPH, implementation of AMTSL, intravenous oxytocin and other estimates compare well with a recently published study using observation measures,48 we believe that the approach we used was acceptable. Moreover, data were consistent during baseline and postintervention leading us to assume that no differential bias was introduced.
PPH was diagnosed by using the kanga method—saturation of the standard-sized local cloth.33 When conceptualising the trial we explored other options such as the blood mat, but the purchase price was beyond our financial capacity—a problem also faced by Nelissen et al.35 This could have led to an under-reporting of PPH cases. However, we believe no differential under-reporting was introduced as intervention and comparison facilities both faced this challenge similarly.
Conclusion
Our large cluster-randomised trial conducted in typical rural districts in Tanzania indicated a significant, although small reduction of PPH near misses after the implementation of the 1-day in-facility HMS BAB training. We saw a positive effect of the training along the implementation line from knowledge and skills to selected implementation practices to reduction of PPH-related near misses and mortality. However, as case fatality is still high, training might need to include additional interventions such as use of tranexamic acid and intrauterine balloon placement. While training is clearly important, it is insufficient on its own to bring the much-needed reduction in PPH near misses and mortality.
Acknowledgments
We thank the Tanzania’s Ministry of Health, Community Development, Gender, Elderly and Children (MoHCDGEC) for guidance and support, regional and district health management teams and administrators and health workers, women and their families for participating in the study. The trial was done as a collaboration between two local professional associations: The Association of Gynaecologists and Obstetricians of Tanzania (AGOTA) and Tanzania Midwifery Association (TAMA). We thank the HMS BAB study team for Tanzania and Uganda: Hilda Kwezi, Lucy Joyce Atim, Shirima Kizito, Dinah Amongin and Nester Moyo of International Confederation of Midwives for all the support and work that went into implementing the trial. Our sincere appreciation to the scientific advisory board members, Professor William Stones, MD, FRCOG, Professor Birgitta Essén, MD, PhD, Associate Professor Hege Ersdal, MD, PhD, and Cherrie Evans, DrPH, CNM, for their oversight and guidance.
References
Footnotes
Handling editor Stephanie M Topp
Contributors CH and FAA conceptualised the study in Tanzania with support from AP, and JM. GM, FAA, AH and CH performed the statistical analysis. FAA, CH, AP and SL were involved into the operationalisation of the study and the data collection support. FAA and CH wrote the first draft of this protocol. All authors contributed to the manuscript development and commented on several drafts of the manuscript. All authors read and approved the final manuscript.
Funding This study was funded by Laerdal Foundation for Acute Medicine (40070).
Competing interests None declared.
Patient consent for publication Not required.
Ethics approval Senate Research and Publication Committee of Muhimbili University of Health and Allied Health Science
Provenance and peer review Not commissioned; externally peer reviewed.
Data sharing statement Additional data will be archived on the Dyrad Digital Repository.