-
PDF
- Split View
-
Views
-
Cite
Cite
L. Simonsen, C. Viboud, A. Elixhauser, R.J. Taylor, A.Z. Kapikian, More on RotaShield and Intussusception: The Role of Age at the Time of Vaccination, The Journal of Infectious Diseases, Volume 192, Issue Supplement_1, September 2005, Pages S36–S43, https://doi.org/10.1086/431512
Close - Share Icon Share
Abstract
RotaShield, a vaccine intended to prevent severe rotavirus diarrhea, was withdrawn in July 1999, 9 months after it became available in the United States, because of a temporal association with intussusception events that occurred in vaccinated infants. We explore here the effect of age on the risk of intussusception.
We reanalyzed a case-control database of the Centers for Disease Control and Prevention by use of a 21-day window, to define vaccine-associated events. We obtained data on vaccine use from the National Immunization Survey and estimated the age-stratified background incidence of intussusception by use of Healthcare Cost and Utilization Project data. We combined these data to estimate how absolute risk varies with age and to model the projected population-attributable risk associated with 3 different vaccination schedules.
We found that the incidence of intussusception associated with the first dose of vaccine increased with age. Infants ⩾90 days old accounted for 80% of cases of intussusception associated with a first dose but had received only 38% of first doses. Modeling of the recommended schedule of vaccination at ages 2, 4, and 6 months projected 1 intussusception event/11,000–16,000 vaccine recipients; modeling of a 2-dose schedule beginning in the neonatal period projected 1 intussusception event/38,000–59,000 vaccine recipients.
The practice of initiating immunization after age 90 days, which we call “catch-up” vaccination, contributed disproportionately to the occurrence of intussusception associated with the use of RotaShield. A fully implemented 2-dose vaccination schedule begun during the neonatal period would lead to, at most, a 7% increase in the incidence of intussusception above the annual background incidence.
Rotaviruses are the most common cause of severe diarrhea among infants and young children and are responsible for significant morbidity and mortality among these age groups worldwide. In the United States, rotavirus infection results in ∼500,000 pediatric outpatient visits, 50,000 hospital admissions, and 20 deaths annually [1]. Worldwide, however, rotaviruses cause ∼600,000 deaths annually among infants and young children, predominantly in developing countries [2].
Beginning in the mid-1980s, Wyeth-Ayerst Pharmaceuticals undertook the development and clinical testing of RotaShield, a live rotavirus vaccine composed of 3 human-rhesus reassortant rotavirus strains and 1 rhesus rotavirus strain. During these trials, 5 intussusception events occurred among 10,054 vaccinated infants; 4 of these events occurred within 3 weeks after administration of the second or third dose [3]. Of these 4 vaccine-associated events, 3 of them occurred in a subset of <2000 infants who were given experimental vaccine formulations that were never marketed (W. Gruber, Wyeth-Ayerst Pharmaceuticals; personal communication). When the US Food and Drug Administration (FDA) licensed RotaShield in August 1998, it required that intussusception be listed in the package insert as a possible adverse event. Distribution began in October 1998, after the Advisory Committee on Immunization Practices (ACIP) of the US Centers for Disease Control and Prevention (CDC) recommended routine immunization of all US infants following a 3-dose schedule, preferably at 2, 4, and 6 months of age [4].
In July 1999, however, the CDC recommended that physicians immediately suspend the use of RotaShield after data collected by the CDC-FDA Vaccine Adverse Events Reporting System revealed a higher-than-expected number of intussusception events among vaccinated infants [5]. Wyeth-Ayerst recalled all unused vaccine doses and withdrew the product from the market, and ACIP withdrew its recommendation.
During the 9 months that RotaShield was in use, ∼1.2 million doses were given to ∼600,000 infants [6, 7]. In October 1999, preliminary estimates suggested that a fully implemented program of RotaShield use would have led to up to 1600 excess intussusception cases, corresponding to a population-attributable risk (PAR) of 1 excess case/2500 vaccine recipients [8]. The results of a multistate, case-control study of vaccinated infants that was conducted by the CDC later confirmed a strong association between receipt of an initial dose of RotaShield and the occurrence of intussusception during the 2 weeks immediately following vaccination, but a lower PAR value (1 excess case of intussusception/4670–9474 infants vaccinated) was estimated [9].
Although estimates of PAR provide a measure of the absolute risk associated with vaccination and are, thus, the most important risk estimates to consider when policy decisions about vaccine use are made, they depend, in part, on measures of relative risk—specifically, assessments of the odds ratio (OR) for intussusception for vaccinated versus unvaccinated infants. The CDC study reported that the OR did not vary with age [9]. We noted, however, that, if the background incidence of intussusception increases during the first months of life, then an unvarying relative risk would yield higher estimates of PAR among infants in the older age groups. Moreover, it seemed to us curious that, in prelicensure trials, the intussusception events among vaccinated infants occurred after receipt of the second or third dose [3], whereas, during the 9-month period of RotaShield use, the first dose was temporally associated with intussusception far more strongly than was the second dose, and the third dose could not be shown to be associated with intussusception at all [9].
We therefore set out to explore more fully the relationship between the age at the time of vaccination and the risk of intussusception. We conducted extended analyses of the case-control database on which the study by Murphy et al. [9] was based and that documented ∼75% of cases of intussusception in the 19-state study area [9]. We identified all cases of intussusception that occurred during the 21 days immediately after vaccination; we took these cases as our set of vaccine-associated cases. Furthermore, we used data from the National Immunization Survey (NIS), to estimate RotaShield vaccination coverage among infants of different ages in these same 19 states, and data from the Healthcare Cost and Utilization Project (HCUP), to estimate age-specific background rates of intussusception in the infant population. We then determined the effect of age at vaccination on both relative and absolute risks of intussusception and estimated the PAR that would be associated with 3 different vaccination schedules.
We found that age at vaccination strongly affects the absolute risk of intussusception. We believe that our results shed new light on the unhappy experience with RotaShield in the United States and can help to maximize the chance that any future rotavirus vaccination programs will succeed.
Methods
Age Designation Convention
The ages of infants are typically expressed in days or months. However, for younger ages in particular, ages expressed in months can be quite imprecise: an infant said to be “2 months of age” can be anywhere from 60 to 89 days old. In the present study, when possible, we express age in days. Ages expressed in months correspond to an age expressed as a range of days, as follows: 0 months, birth to 29 days; 1 month, 30–59 days; and so forth (table 1).
Incidence of intussusception associated with administration of the first dose of RotaShield and no. of first doses received, by age, in 19 US states during the 9-month period of RotaShield use, 1998–1999.
| Group, age at first dose of RotaShield | No. of infants with intussusception (% of all intussusception events)a | No. of first doses given (% of infants vaccinated)b | Incidence of intussusception/100,000 first doses |
| Younger infants | |||
| <30 days (0 months) | 0 | 1935 | 0 |
| 30–59 days (1 month) | 0 | 69,123 | 0 |
| 60–89 days (2 months) | 9 | 197,144 | 5 |
| <90 days (⩽2 months) | 9 (20) | 268,202 (62) | 3 |
| Older infants | |||
| 90–119 days (3 months) | 6 | 35,441 | 17 |
| 120–159 days (4 months) | 17 | 77,413 | 22 |
| 160–189 days (5 months) | 2 | 15,088 | 13 |
| 190–209 (6 months) | 11 | 32,534 | 34 |
| 210–239 days (7 months | 0 | 779 | 0 |
| 240–269 days (8 months) | 0 | 868 | 0 |
| 270–299 days (9 months) | 0 | 2896 | 0 |
| 300–329 days (10 months) | 0 | 0 | 0 |
| 330–365 days (11 months) | 0 | 0 | 0 |
| ⩾90 days (>2 months) | 36 (80) | 165,019 (38) | 22 |
| All (<365 days, <12 months) | 45 (100) | 433,222 (100) | 10 |
| Group, age at first dose of RotaShield | No. of infants with intussusception (% of all intussusception events)a | No. of first doses given (% of infants vaccinated)b | Incidence of intussusception/100,000 first doses |
| Younger infants | |||
| <30 days (0 months) | 0 | 1935 | 0 |
| 30–59 days (1 month) | 0 | 69,123 | 0 |
| 60–89 days (2 months) | 9 | 197,144 | 5 |
| <90 days (⩽2 months) | 9 (20) | 268,202 (62) | 3 |
| Older infants | |||
| 90–119 days (3 months) | 6 | 35,441 | 17 |
| 120–159 days (4 months) | 17 | 77,413 | 22 |
| 160–189 days (5 months) | 2 | 15,088 | 13 |
| 190–209 (6 months) | 11 | 32,534 | 34 |
| 210–239 days (7 months | 0 | 779 | 0 |
| 240–269 days (8 months) | 0 | 868 | 0 |
| 270–299 days (9 months) | 0 | 2896 | 0 |
| 300–329 days (10 months) | 0 | 0 | 0 |
| 330–365 days (11 months) | 0 | 0 | 0 |
| ⩾90 days (>2 months) | 36 (80) | 165,019 (38) | 22 |
| All (<365 days, <12 months) | 45 (100) | 433,222 (100) | 10 |
Data are from a case-control study [9]. This table underestimates the true incidence, because the case-control study reported only ∼75% of all cases of intussusception that occurred in the 19 states. More important, the relative differences in age-specific incidences are accurate.
Data are from the National Immunization Survey conducted in 19 states, as analyzed in the present study.
Incidence of intussusception associated with administration of the first dose of RotaShield and no. of first doses received, by age, in 19 US states during the 9-month period of RotaShield use, 1998–1999.
| Group, age at first dose of RotaShield | No. of infants with intussusception (% of all intussusception events)a | No. of first doses given (% of infants vaccinated)b | Incidence of intussusception/100,000 first doses |
| Younger infants | |||
| <30 days (0 months) | 0 | 1935 | 0 |
| 30–59 days (1 month) | 0 | 69,123 | 0 |
| 60–89 days (2 months) | 9 | 197,144 | 5 |
| <90 days (⩽2 months) | 9 (20) | 268,202 (62) | 3 |
| Older infants | |||
| 90–119 days (3 months) | 6 | 35,441 | 17 |
| 120–159 days (4 months) | 17 | 77,413 | 22 |
| 160–189 days (5 months) | 2 | 15,088 | 13 |
| 190–209 (6 months) | 11 | 32,534 | 34 |
| 210–239 days (7 months | 0 | 779 | 0 |
| 240–269 days (8 months) | 0 | 868 | 0 |
| 270–299 days (9 months) | 0 | 2896 | 0 |
| 300–329 days (10 months) | 0 | 0 | 0 |
| 330–365 days (11 months) | 0 | 0 | 0 |
| ⩾90 days (>2 months) | 36 (80) | 165,019 (38) | 22 |
| All (<365 days, <12 months) | 45 (100) | 433,222 (100) | 10 |
| Group, age at first dose of RotaShield | No. of infants with intussusception (% of all intussusception events)a | No. of first doses given (% of infants vaccinated)b | Incidence of intussusception/100,000 first doses |
| Younger infants | |||
| <30 days (0 months) | 0 | 1935 | 0 |
| 30–59 days (1 month) | 0 | 69,123 | 0 |
| 60–89 days (2 months) | 9 | 197,144 | 5 |
| <90 days (⩽2 months) | 9 (20) | 268,202 (62) | 3 |
| Older infants | |||
| 90–119 days (3 months) | 6 | 35,441 | 17 |
| 120–159 days (4 months) | 17 | 77,413 | 22 |
| 160–189 days (5 months) | 2 | 15,088 | 13 |
| 190–209 (6 months) | 11 | 32,534 | 34 |
| 210–239 days (7 months | 0 | 779 | 0 |
| 240–269 days (8 months) | 0 | 868 | 0 |
| 270–299 days (9 months) | 0 | 2896 | 0 |
| 300–329 days (10 months) | 0 | 0 | 0 |
| 330–365 days (11 months) | 0 | 0 | 0 |
| ⩾90 days (>2 months) | 36 (80) | 165,019 (38) | 22 |
| All (<365 days, <12 months) | 45 (100) | 433,222 (100) | 10 |
Data are from a case-control study [9]. This table underestimates the true incidence, because the case-control study reported only ∼75% of all cases of intussusception that occurred in the 19 states. More important, the relative differences in age-specific incidences are accurate.
Data are from the National Immunization Survey conducted in 19 states, as analyzed in the present study.
Data Sources
Case-control study
The CDC made available to us the case-control study database on which their original findings were based [9], and we used the database to explore more fully the role that the age of the infant plays in the risk of intussusception. This database was constructed through exhaustive hospital-based surveillance and verification of intussusception cases from large hospitals in 19 states in which RotaShield was known to be heavily used (CA, GA, IL, IN, MD, MI, MN, MO, NE, NJ, NY, NC, OH, PA, SC, TN, TX, VA, and WI) [9]. This study identified 433 intussusception events, and it was estimated that this number represented ∼75% of all intussusception events that occurred in these 19 states during the study period. We extended the analysis of these 433 events, paying particular attention to variables identifying age at vaccination, calendar month of hospitalization for intussusception, and age at which each infant received each dose of the vaccine. We used the original conditional logistic regression models generated by the CDC [9] to estimate the adjusted ORs for intussusception associated with exposure to RotaShield within 21 days before the onset of intussusception (or before the reference date for control subjects).
NIS database
Data from the NIS [10] were analyzed and made available to us by Meena Khare (National Center for Health Statistics, CDC). These data provided information from interviews of parents of infants about exposure to RotaShield. These data were used to generate population-based estimates of the age of children at RotaShield vaccine uptake, both by dose and over time during the period of RotaShield use. A total of 13,778 children born from March 1998 through May 1999 were interviewed in the 19 states included in the case-control study. Weighted estimates of the number and age distribution of vaccinated infants in the 19 states were generated using the Sudaan statistical package (version 7; Statistical Center).
HCUP database
Data from the HCUP State Inpatient Databases provided information obtained at discharge from a near-census of inpatients discharged from hospitals in 22 states (AZ, CA, CT, CO, FL, GA, HI, IA, IL, KS, MA, MD, MO, NJ, NY, OR, PA, SC, TN, UT, WA, and WI), including ∼2.4 million infants (i.e., >60% of the annual US birth cohort). Data on age were expressed in both days and months for the 3 years flanking the period of RotaShield use (i.e., 1997, 1998, and 2000). For these 3 years, we extracted data from records of all hospitalizations that made any mention of intussusception (International Classification of Diseases, Ninth Revision, Clinical Modification, code 560.0). We estimated age-specific incidence by month of age using state birth cohort data for 1998, with the assumption that equal numbers of infants were born each month (detailed methods are described elsewhere [11]). Because HCUP data were available for only 11 of the 19 states included in the CDC case-control study, we elected to use data from all 22 states included in the HCUP database, to generate age-specific background incidence rates.
Statistical Analyses: Model Used to Project PAR
Relative risks (i.e., odds ratios [ORs]) of the occurrence of RotaShield-associated intussusception within 21 days after administration of a dose, by dose and age.
| Age, months (days) | First dose | Second dose | Third dose |
| 1–2 (30–89) | 5.7 (1.2–28.3) | …a | …a |
| 3–4 (90–149) | 10.5 (4.0–27.4) | 3.5 (0.98–12.3) | …a |
| 5–11 (150–365) | 15.9 (4.6–54.2) | 0.3 (0.03–2.5) | 0.6 (0.1–4.2) |
| 1–11 (30–365) | 8.6 (4.6–16) | 1.7 (0.7–4.4) | 0.8 (0.2–4.6) |
| Age, months (days) | First dose | Second dose | Third dose |
| 1–2 (30–89) | 5.7 (1.2–28.3) | …a | …a |
| 3–4 (90–149) | 10.5 (4.0–27.4) | 3.5 (0.98–12.3) | …a |
| 5–11 (150–365) | 15.9 (4.6–54.2) | 0.3 (0.03–2.5) | 0.6 (0.1–4.2) |
| 1–11 (30–365) | 8.6 (4.6–16) | 1.7 (0.7–4.4) | 0.8 (0.2–4.6) |
NOTE. Data are ORs (95% confidence intervals). Matched, socioeconomic status-adjusted OR estimates were calculated by use of a conditional regression model, as described elsewhere [4], but with stratification by age (at referent date) and consideration of a 0–21-day exposure window. Data are derived from Murphy et al. [9].
The OR was 0 or indefinite, because there were no or a small number of exposed case patients and/or control subjects.
Relative risks (i.e., odds ratios [ORs]) of the occurrence of RotaShield-associated intussusception within 21 days after administration of a dose, by dose and age.
| Age, months (days) | First dose | Second dose | Third dose |
| 1–2 (30–89) | 5.7 (1.2–28.3) | …a | …a |
| 3–4 (90–149) | 10.5 (4.0–27.4) | 3.5 (0.98–12.3) | …a |
| 5–11 (150–365) | 15.9 (4.6–54.2) | 0.3 (0.03–2.5) | 0.6 (0.1–4.2) |
| 1–11 (30–365) | 8.6 (4.6–16) | 1.7 (0.7–4.4) | 0.8 (0.2–4.6) |
| Age, months (days) | First dose | Second dose | Third dose |
| 1–2 (30–89) | 5.7 (1.2–28.3) | …a | …a |
| 3–4 (90–149) | 10.5 (4.0–27.4) | 3.5 (0.98–12.3) | …a |
| 5–11 (150–365) | 15.9 (4.6–54.2) | 0.3 (0.03–2.5) | 0.6 (0.1–4.2) |
| 1–11 (30–365) | 8.6 (4.6–16) | 1.7 (0.7–4.4) | 0.8 (0.2–4.6) |
NOTE. Data are ORs (95% confidence intervals). Matched, socioeconomic status-adjusted OR estimates were calculated by use of a conditional regression model, as described elsewhere [4], but with stratification by age (at referent date) and consideration of a 0–21-day exposure window. Data are derived from Murphy et al. [9].
The OR was 0 or indefinite, because there were no or a small number of exposed case patients and/or control subjects.
Results
Age and Intussusception: Highest Risk for “Catch-Up” Infants
Relative risk of intussusception (CDC case-control study)
For all ages combined, our adjusted OR estimates were 8.6 for the first dose, 1.7 for the second dose, and 0.8 for the third dose (table 2). We note that a relative risk of 8.6 would suggest that ∼90% of intussusception events that occurred 0–21 days after administration of the first dose were attributable to the vaccine, and that the remaining 10% of events were background events.
When we stratified the intussusception cases into 3 groups by age (i.e., ⩽2, 3–4, and ⩾5 months), we found that the adjusted OR point estimates for the first dose increased with age, but with wide and overlapping confidence intervals (table 2). It was not possible to specifically study the relative risk for the youngest age groups, because no RotaShield-associated cases of intussusception recorded in the database occurred among infants <60 days old. for the second dose, there was a tendency toward a higher relative risk for the younger age group (3–4 months) than for the older age group (⩾5 months), but, with only 9 cases associated with a second dose, the confidence intervals were wide.
Age at vaccination for all vaccinated infants (NIS data)
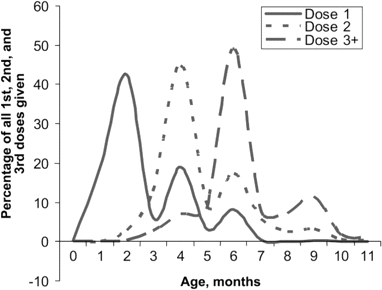
The mean age at receipt of RotaShield vaccine was 97 days (3 months) for the first dose, 155 days (5 months) for the second dose, and 211 days (7 months) for the third dose. Figure 1 shows the percentage of the total number of first, second, and third doses of RotaShield given, at various ages, to the population of infants in the 19 states included in the CDC database. The prominent peaks occurring at ages 2, 4, and 6 months in association with administration of the first, second, and third doses, respectively, show that approximately one-half of the infants received RotaShield according to the target schedule given by ACIP and used in clinical testing. Specifically, 62% of vaccinated infants in the 19 states received the first dose at <90 days of age. Furthermore, the fact that 38% of vaccinated infants received the first dose at ⩾90 days of age indicates that a substantial minority of infants vaccinated were “catch-up” infants—that is, older infants who began receiving the RotaShield dose series in an effort to allow them to “catch up” with the schedule and, therefore, still benefit from the vaccine. Finally, 16% of vaccinated infants (∼70,000 infants in the 19 states) received the first dose at <60 days of age, indicating that early vaccination also occurred (table 1).
Age at receipt of first, second, and third doses of RotaShield among US infants, October 1998–July 1999. Data are from the National Immunization Survey, which covered 19 states.
Age at vaccination for infants with RotaShield-associated intussusception (case-control and NIS data combined)
The CDC case-control study database identified 57 RotaShield-associated intussusception events that occurred within 21 days of receipt of a dose; 45 of these events occurred after receipt of a first dose, 10 occurred after receipt of a second dose, and 2 occurred after receipt of a third dose. We note that intussusception events occurred disproportionately among older infants: only 9 (20%) of the 45 infants who developed intussusception within 21 days after receipt of a first dose were <90 days of age at vaccination, even though this age group received 62% of first vaccine doses (P < .0001). We also note that no case of intussusception occurred among infants vaccinated at <60 days of age, even though this age group received 16% of all first vaccination doses.
Incidence of RotaShield-associated intussusception by age
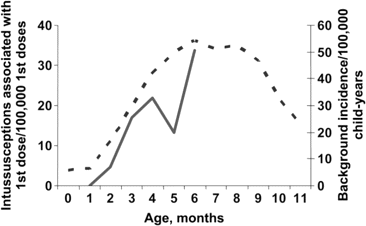
The observed incidence of intussusception per 100,000 first doses in the 19 states included in the CDC study increased with age (figure 2). Because the CDC database did not document all the intussusception events that occurred in the states surveyed, the estimates of incidence here are understated; however, because there is no indication of any age bias in the tally of intussusception events, the increasing trend with age shown in figure 2 should be accurate. We also note that the increase in the incidence of intussusception with age during the period of RotaShield use closely mirrors the increase that occurs with age in the background incidence of intussusception (figure 2).
Observed age-specific incidence of intussusception associated with the use of RotaShield (solid line) and annual background incidence of intussusception in the United States estimated from Healthcare Cost and Utilization Project data (dotted line).
Background incidence of intussusception
Data from the 22 states included in the HCUP database indicate that the annual background incidence of intussusception was 35.7 intussusceptions/100,000 infants; this value was age-proportionally adjusted to 34.2 intussusceptions/100,000 infants, to match the annual incidence used in the study by Murphy et al. [9]. Our HCUP-based estimates of the age-specific background incidence of intussusception increased by ∼10-fold, from ∼5 to ∼50 intussusceptions/100,000 infants during the first 6 months of life (figure 2).
Deferred risk of intussusception associated with second and third doses
Of the 57 intussusception events that were temporally associated with RotaShield in the case-control study, only 10 were associated with receipt of a second RotaShield dose [9]. We note that these 10 infants were, on average, considerably younger (median age, 67.5 days) when they received the first dose than were those who experienced intussusception after receipt of a first dose (median age, 130 days) (P = .008, Wilcoxon test). Moreover, 8 of the 10 infants with intussusception related to a second dose were <90 days old when they received their first dose, compared with 9 of 45 infants with intussusception related to a first dose (P < .001). in addition, our finding that estimates of the OR for the second dose tend to decrease with age (table 2) supports the possibility that some of the cases of intussusception related to receipt of a second dose might have been deferred—that is, that the events occurred in infants who were prone to intussusception but did not occur until after the infants had reached the age at which the risk of intussusception is higher and had received their second dose of vaccine. This possibility could not be further explored or quantified with the data available.
Modeling of PAR According to 3 Vaccination Schedules
We first used the background age-stratified estimates of incidence of intussusception in combination with the dose-specific estimates of ORs to project the PAR according to 3 different fully implemented vaccination schedules (table 3). We then introduced age-specific measures of the relative risk associated with receipt of the first dose into the model, using our OR point estimates from different age ranges (table 2). Furthermore, because the OR for the third dose was always <1, we assumed that there were no excess cases associated with receipt of the third dose.
Model of projected absolute risk of intussusception attributable to RotaShield for 3 weeks after immunization, in a fully (90%) immunized annual US birth cohort of 3.4 million infants, under 3 different vaccination schedules.
| Outcome | Standard schedule (2, 4, and 6 months) | “Catch-up” schedule (3, 5, and 7 months)a | Neonatal schedule (2 doses at 0 and 1 months) |
| No. of associated intussusception cases after administration of: | |||
| First dose | 155b–250c | 553c–797b | 50b–81c |
| Second dosed | 62 | 73 | 9 |
| Third dosee | 0 | 0 | 0 |
| Schedule total | 217b–312c | 625c–869b | 59b–90c |
| Projected PAR, no. of vaccine recipients per each excess intussusception | 11,000c–16,000b | 5500b–6700c | 38,000c–58,000b |
| Elevation above annual background incidence of intussusception events, %f | 17b–24c | 40c–48b | 5b–7c |
| Outcome | Standard schedule (2, 4, and 6 months) | “Catch-up” schedule (3, 5, and 7 months)a | Neonatal schedule (2 doses at 0 and 1 months) |
| No. of associated intussusception cases after administration of: | |||
| First dose | 155b–250c | 553c–797b | 50b–81c |
| Second dosed | 62 | 73 | 9 |
| Third dosee | 0 | 0 | 0 |
| Schedule total | 217b–312c | 625c–869b | 59b–90c |
| Projected PAR, no. of vaccine recipients per each excess intussusception | 11,000c–16,000b | 5500b–6700c | 38,000c–58,000b |
| Elevation above annual background incidence of intussusception events, %f | 17b–24c | 40c–48b | 5b–7c |
NOTE. PAR, population-attributable risk.
Shown are the actual mean ages of the infants during the total 9-month period of RotaShield use.
Using actual point-estimate of odds ratio (OR), by age (see table 2).
Using unchanging relative risk, by age (OR, 8.6).
For the second dose, estimates of absolute risk were calculated under the assumption that the OR did not change on the basis of age (OR, 1.7); the possibility of a “deferred” risk of a second dose was not taken into account when we calculated these estimates.
For the third dose, we assumed that there were no RotaShield-associated cases, because the OR was estimated to be 0.8 (see table 2).
Estimated annual average no. of intussusception cases among US infants <1 year old is 1291.
Model of projected absolute risk of intussusception attributable to RotaShield for 3 weeks after immunization, in a fully (90%) immunized annual US birth cohort of 3.4 million infants, under 3 different vaccination schedules.
| Outcome | Standard schedule (2, 4, and 6 months) | “Catch-up” schedule (3, 5, and 7 months)a | Neonatal schedule (2 doses at 0 and 1 months) |
| No. of associated intussusception cases after administration of: | |||
| First dose | 155b–250c | 553c–797b | 50b–81c |
| Second dosed | 62 | 73 | 9 |
| Third dosee | 0 | 0 | 0 |
| Schedule total | 217b–312c | 625c–869b | 59b–90c |
| Projected PAR, no. of vaccine recipients per each excess intussusception | 11,000c–16,000b | 5500b–6700c | 38,000c–58,000b |
| Elevation above annual background incidence of intussusception events, %f | 17b–24c | 40c–48b | 5b–7c |
| Outcome | Standard schedule (2, 4, and 6 months) | “Catch-up” schedule (3, 5, and 7 months)a | Neonatal schedule (2 doses at 0 and 1 months) |
| No. of associated intussusception cases after administration of: | |||
| First dose | 155b–250c | 553c–797b | 50b–81c |
| Second dosed | 62 | 73 | 9 |
| Third dosee | 0 | 0 | 0 |
| Schedule total | 217b–312c | 625c–869b | 59b–90c |
| Projected PAR, no. of vaccine recipients per each excess intussusception | 11,000c–16,000b | 5500b–6700c | 38,000c–58,000b |
| Elevation above annual background incidence of intussusception events, %f | 17b–24c | 40c–48b | 5b–7c |
NOTE. PAR, population-attributable risk.
Shown are the actual mean ages of the infants during the total 9-month period of RotaShield use.
Using actual point-estimate of odds ratio (OR), by age (see table 2).
Using unchanging relative risk, by age (OR, 8.6).
For the second dose, estimates of absolute risk were calculated under the assumption that the OR did not change on the basis of age (OR, 1.7); the possibility of a “deferred” risk of a second dose was not taken into account when we calculated these estimates.
For the third dose, we assumed that there were no RotaShield-associated cases, because the OR was estimated to be 0.8 (see table 2).
Estimated annual average no. of intussusception cases among US infants <1 year old is 1291.
Under the assumption that the relative risk did not change with age, the model projected an estimate of 1 excess intussusception event/11,000 vaccine recipients (corresponding to 312 excess cases) when the standard 2-, 4-, and 6-month dosing schedule was used. The model projected considerably more intussusception events in association with the catch-up scenario (PAR, 1 in 6700 vaccine recipients), but many fewer such events in association with the neonatal scenario (PAR, 1 in 38,000 vaccine recipients). Under the assumption that the relative risk associated with administration of the first dose changed with age, the model projected fewer excess cases in association with the neonatal and standard scenario and more in association with the catch-up scenario (table 3).
Discussion
We have previously argued that most intussusception events associated with the use of RotaShield would likely have occurred at some point during infancy, even without exposure to RotaShield, and that receipt of the vaccine, therefore, did not change the overall risk of intussusception during the first year of life [11, 12] (for other interpretations, however, see the studies by Murphy et al. [13, 14]). Nonetheless, the strong temporal association between intussusception and receipt of RotaShield represents a serious problem that must be addressed.
Effect of age on the absolute risk of intussusception after exposure to RotaShield
Our analysis shows conclusively that the number of intussusception events associated with receipt of a first dose of RotaShield vaccine—which we defined as any intussusception event that occurred within 21 days of receipt of a vaccine dose—increases with age. This finding was supported both by observational data showing an increased incidence of intussusception events associated with receipt of the first dose with increased age (table 2; figure 2) and by our modeling results (table 3).
The increase in absolute risk associated with age was largely due to an increase in the background rate of intussusception with age during the first 6 months of infancy. The relative risk was high enough that almost all intussusception events that occurred during the 21 days immediately after receipt of a first RotaShield dose were attributable to the vaccine. However, beyond the effect of the increased background incidence, our finding that the relative risk after receipt of a first dose increases with age further accentuated the effect of age on our projections of PAR. Our model estimated a PAR of 1 excess case of intussusception/11,000–16,000 vaccine recipients for the standard schedule of dosing at 2, 4, and 6 months, a result similar to the PAR of 1 excess case/9474 vaccine recipients estimated by Murphy et al. [9] by use of the same data, under the assumption that the OR did not change with age. Because the background incidence increases with age, the number of cases associated with receipt of the vaccine increases when infants begin the vaccine series at later ages. For example, our model projected that a fully implemented catch-up schedule of doses administered at ages 3, 5, and 7 months would produce 553–797 additional cases (PAR, 1 excess case/5500–6700 vaccine recipients), for a 40%–48% increase over the annual background incidence. Our modeling results also indicate that a 2-dose neonatal schedule of vaccination, administered at ages 0–29 and 30–59 days, would produce far fewer intussusception events than would the standard schedule; specifically, we project that this schedule would be associated with 59–81 excess cases (PAR, 1 excess case/38,000–58,000 vaccine recipients), which is a 5%–7% increase above the annual background incidence (table 2). Such a small increase would be difficult to detect in a fully implemented vaccination program and would be well within the range of the risk of severe adverse effects associated with other vaccines, such as that against measles, mumps, and rubella [15, 16]. These estimates are consistent with the observation that no vaccine-associated cases of intussusception were observed among infants <60 days old, although, in the study of 19 states, infants in this age group had received 70,000 first doses.
Deferred risk of intussusception
We found that infants who developed intussusception after receipt of a second dose had started their vaccination series when they were substantially younger than infants who experienced intussusception after receipt of a first dose. We, therefore, suggest that many of the cases associated with a second dose had been simply “deferred,” in that they occurred among infants who were prone to intussusception but who were very young at receipt of the first dose, and were, therefore, unlikely to experience intussusception then. This concept also explains the curious pattern seen in the RotaShield trials, in which all 4 of the intussusception events that were observed occurred after receipt of a second or third dose [3]. Because infants in the trials were vaccinated on a rigorous 2-, 4-, and 6-month schedule with no catch-up immunization, they were young enough at the time of administration of the first dose that the occurrence of intussusception was likely deferred until after receipt of the second or third dose.
Implications for future use of rotavirus vaccines
Although statistical evidence indicates that exposure to RotaShield caused intussusception, no biological mechanism has been established. Consequently, it is difficult to say whether intussusception was a problem unique to RotaShield or whether it will affect other live rotavirus vaccines. One recent study of 13 pairs of infants hospitalized with rotavirus diarrhea documented a spontaneously reversed intussusception event, as well as significant anatomic small-bowel changes and lymph-node aggregates, suggesting that natural rotavirus infections may be a prelude to intussusception [17]. By extension, then, any live rotavirus vaccine candidate may carry with it a risk of intussusception.
In the wake of the continuing debate about the magnitude of the risk of intussusception, efforts are currently in progress to bring back RotaShield to the international market [18, 19]. We have demonstrated that a neonatal vaccination schedule would greatly reduce the risk of intussusception. As an added benefit, data from a clinical trial of RotaShield conducted in Finland [20] suggest that fever, which is commonly associated with RotaShield use [21], might be avoided under a neonatal vaccination schedule (Timo Vesikari, unpublished data). The efficacy of neonatal administration of the vaccine would need to be established in clinical trials. Asymptomatic neonatal infection has been shown to protect against severe rotavirus diarrhea in Australia [22], and, in Finland, vaccination with a neonatal bovine rotavirus (RIT 4237) reduced the severity of subsequent episodes of gastroenteritis due to rotavirus infection [23].
Currently, 2 second-generation live rotavirus vaccines manufactured by Merck [24] and GlaxoSmithKline [25] are undergoing phase 3 trials. Given our findings that age has an effect on the risk of intussusception, and given the difficulty of demonstrating that new rotavirus vaccines do not cause such a rare event as intussusception in prelicensure trials, we offer 3 suggestions for the future. First, it would be prudent to avoid catch-up immunization, so that the first dose is completed by age 60 days, before infants reach the age at which the background incidence of intussusception begins to increase. Second, because of the unresolved question regarding a “deferred” risk of intussusception associated with receipt of a second or third dose, completion of the series before age 60 days might be even more prudent; this would require limiting the series to 2 doses. Third, regulatory agencies should decide in advance of introductions of rotavirus vaccines what level of intussusception risk is acceptable, bearing in mind that an effective rotavirus vaccine could prevent hundreds of thousands of deaths among children annually worldwide [26].
Acknowledgments
We thank Meena Khare (Centers for Disease Control and Prevention/National Immunization Program, Atlanta, GA), for providing the National Immunization Survey RotaShield coverage data needed for this study; Murphy et al. [9], for making their case-control study database available to us; the statewide data organizations in Arizona, California, Colorado, Connecticut, Florida, Georgia, Hawaii, Illinois, Iowa, Kansas, Maryland, Massachusetts, Missouri, New Jersey, New York, Oregon, Pennsylvania, South Carolina, Tennessee, Utah, Washington, and Wisconsin, for contributing data to the Healthcare Cost and Utilization Project State Inpatient Databases used in this study; and Ken Rothman (Boston University) and Bill Blackwelder, Dean Follman, John La Montagne, David Morens, and Brian Murphy (National Institutes of Health), for helpful suggestions.
References
This article is in the public domain, and no copyright is claimed.
Author notes
Presented in part: 6th International Rotavirus Symposium, 7–9 July 2004, Mexico City, Mexico.
Potential conflicts of interest: none reported.
Contractor to National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, Maryland.
Reprints or correspondence: Dr. Lone Simonsen, Office of Global Affairs, National Institute of Allergy and Infectious Diseases, National Institutes of Health, 6610 Rockledge Dr., Rm. 2033, Bethesda, MD 20892-6613 (Lsimonsen@niaid.nih.gov).