Abstract
The goal of the present study was to assess the evolution of the in vitro chloroquine resistance and also the prevalence of pfcrt T76 and pfmdr1 Y86 mutations in Pikine from 2000 while chloroquine (CQ) was the first-line treatment of malaria to 2009 when artemisinin-based combination therapies (ACTs) are in use. We genotyped pfcrt K76T and pfmdr1 N86Y polymorphisms by PCR-RFLP and assessed in vitro CQ susceptibility by double-site enzyme-linked pLDH immunodetection (DELI) assay in Plasmodium falciparum isolates collected in Pikine, Senegal. The proportions of the pfcrt T76 allele in the light of the three different treatment policies were 72.4 % before CQ withdrawal (2000 to 2003), 47.2 % while amodiaquine plus Fansidar was the first-line treatment (2004 to 2005), and 59.5 % since the ACT use was implemented (2006 to 2009). The prevalence of pfcrt T76 decreased significantly after CQ was stopped [X 2 = 6.54, P = 0.01 (2000–2003 versus 2004–2005)] and then slightly since ACTs have been implemented [X 2 = 1.12, P = 0.28 (2000–2003 versus 2006–2009)]. There were no significant differences on the prevalence of pfmdr1 Y86 throughout the three treatment policies. The DELI assay was carried out episodically in 2000 (n = 36), 2001 (n = 47), and 2009 (n = 37). The mean IC50s of the isolates to CQ in 2000 versus 2009 and 2001 versus 2009 are significantly different (P < 0.05). The Fisher exact test found a significant association between the presence of the pfcrt T76 mutant allele and in vitro resistance in 2000/2001 (P = 0.023), while in 2009 there were no association between both variables (P = 0.274). Mutant pfcrt T76 and pfmdr1 Y86 alleles and in vitro CQ-resistant strains are still circulating in Pikine. The official discontinuation of CQ use is not completely followed by its total withdrawal from private drug sellers, and the molecule still exerts pressure on local P. falciparum populations.
Similar content being viewed by others
Introduction
In Senegal, chloroquine (CQ) was for decades the first-line treatment for falciparum malaria in Senegal, and the first case of CQ treatment failure was recorded in Dakar, the capital city in 1988 (Trape et al. 1989) before spreading in remote regions (Gaye et al. 1990, 1993; Sokhna et al. 1997). Despite the emergence and spread of chloroquine resistance (CQR), a 3-day CQ course (25 mg/kg) was maintained as treatment of malaria infection until 2003. Then treatment policy was modified by a combination of amodiaquine plus sulfadoxine–pyrimethamine, which in turn was replaced in 2006 by two artemisinin-based combination therapies (ACTs) with either artesunate–amodiaquine or artemether–lumefantrine. To date, the ACTs have demonstrated good efficacy and tolerability (Faye et al. 2007) and this finding is also evidenced by the significant decrease of malaria-attributed deaths (WHO 2010) within the country. However the identification of large ranges of artesunate IC50 values in isolates from Thies in Senegal (Ndiaye et al. 2010) and the first reports showing reduced in vivo susceptibility to artesunate in Cambodia (Alker et al. 2007; Dondorp et al. 2009; Noedl et al. 2009) have prompted our group and the National Malaria Control Program to assess first the prevalence of Plasmodium falciparum chloroquine resistance transporter (pfcrt) T76 and P. falciparum multidrug resistance-1 (pfmdr1) Y86 markers and second the in vitro efficacy of CQ even though the clinical efficacy of the ACT has not yet been compromised.
The P. falciparum transporter genes pfcrt and pfmrdr-1 have been linked to CQR (Reed et al. 2000; Fidock et al. 2000). The mechanisms underlying CQR have been studied extensively, and the accepted mechanism has been the reduced accumulation of CQ in P. falciparum-resistant strains (Krogstad et al. 1987). It is admitted that transporters such as pfcrt and pfmdr1 constitute mediators of CQR in P. falciparum malaria infection.
Interestingly, in many areas of sub-Saharan Africa and Asia (Schwenke et al. 2001; Mita et al. 2003; Kublin et al. 2003; Mwai et al. 2009; Wang et al. 2005) where CQ has been abandoned because of CQR, a reduction in resistance to the molecule has been noticed. This phenomenon has raised hopes about its reintroduction (Laufer et al. 2006). Our study aimed to assess the evolution of the in vitro CQR and also the prevalence of pfcrt T76 and pfmdr1 Y86 mutations in Pikine from 2000 to 2009 and discuss the reintroduction of CQ alone or in combination with artesunate.
Materials and methods
Study site and study population
The study was conducted in an outpatient clinic situated in Pikine a suburb of Dakar and patients with peripheral blood smears positive for P. falciparum, and non-complicated malaria, aged 5 years or greater were invited to participate in the study as previously reported (Thomas et al. 2002; Sarr et al. 2005, 2008). They were excluded in case of complicated malaria, pregnancy, and recent history of antimalarial treatment. Additionally in 2000 and 2001, we performed CQ metabolite checks in patients’ urine, excluding those with positive results. We collected 5 to 10 ml of venous blood from the study participants. The study was approved by the IRBs in Senegal and the Harvard School of Public Health in Boston.
In vitro chloroquine susceptibility test
We used the double-site enzyme-linked pLDH immunodetection (DELI) assay to estimate the CQ susceptibility of the isolates collected from the patients. Two cultured-adapted strains of P. falciparum were used as reference: the CQ-resistant strain W2 and the CQ-susceptible strain 3D7. The in vitro susceptibility to CQ was carried out in duplicate on all the isolates according to procedures described elsewhere (Moreno et al. 2001; Thomas et al. 2002). Briefly, infected erythrocytes were diluted in RPMI 1640 medium supplemented with Albumax (Gibco BRL, Grand Island, NY) and 1 mg/ml of hypoxanthine (Sigma, St. Louis, MO) to obtain a hematocrit of 2 % and a parasitemia of 0.5 to 1 %. Then 200 μl aliquots of the resulting dilutions was individually added to microplate wells precoated with serial dilutions of CQ sulfate (Rhone Poulen Rorer Vitry, France). The plates were incubated in candle jars for 48 h at 37 °C. The isolates with IC50 greater than 100 nM were considered as resistant.
Genotyping
From 2000 through 2006, parasite DNA was extracted from whole blood as described elsewhere (Basco and Ringwald 2000). However in 2007 and 2009, we extracted it from Whatman FTA Classic Card using the QIAGEN Kit according to the manufacturer’s instructions. The two codons of interest pfcrt K76T and pfmdr1 N86Y were amplified with methods described elsewhere (Djimde et al. 2001; Duraisingh et al. 1997). RFLPs were performed on pfcrt K76T and pfmd1 N86Y with Apo I and Afl III, respectively.
Statistical analysis
Statistical analyses were performed using STATA 9 software (Stata Corporation, College Station, TX, USA). Samples carrying both mutant and wild-type forms were excluded from the analysis. The mean IC50s was compared by t tests. Statistical associations were calculated using the Fisher’s exact test and statistical significance was set on an alpha level of 0.05.
Results
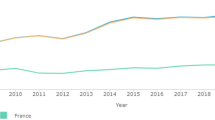
In total, 532 samples were genotyped for pfcrt K76T and pfmdr1 N86Y polymorphisms ranging from 34 in 2000, 53, 79, 70, 79, 98, 82, and 37, in 2001, 2002, 2003, 2004, 2005, 2006, and 2009, respectively. Figure 1 summarizes the evolution of the proportions of pfcrt T76 and pfmdr1 Y86 from 2000 through 2009.
The pfcrt T76 allele distribution presented a surprising pattern. The different proportions in the light of the three different treatment policies were 72.4 % before CQ withdrawal’s (2000 to 2003), 47.16 % while amodiaquine plus fansidar was the first-line treatment (2004 to 2005), and 59.46 % since the ACTs were implemented (2006 to 2009). The prevalence of pfcrt T76 decreased significantly after CQ was stopped [X 2 = 6.54, P = 0.01 (2000–2003 versus 2004–2005)], and then increased slightly since ACTs have been implemented [X 2 = 1.12, P = 0.28 (2000–2003 versus 2006–2009)].
The prevalence of the pfmdr1 Y86 mutant allele almost paralleled the pfcrt T76 trend, although from 2005 to 2009, the proportion of isolates harboring this allele has stabilized. There were no significant differences on the prevalence of pfmdr1 Y86 throughout the three treatment policies.
The DELI assay was carried out episodically in 2000 (n = 36), 2001 (n = 47), and 2009 (n = 37). The mean IC50s in 2000 versus 2009 and 2001 versus 2009 are significantly different (P < 0.05). The Fisher exact test found a significant association between the presence of the pfcrt T76 allele and in vitro resistance in 2000/2001 (P = 0.023), while in 2009 there were no association between both variables (P = 0.274).
Discussion
Unlike the trends observed in Malawi, Kenya, Gabon, and China (Schwenke et al. 2001; Mita et al. 2003; Kublin et al. 2003; Mwai et al. 2009; Wang et al. 2005) where a constant drop of the pfcrt T76 prevalence has been noticed, our finding is in favor of a surprising pattern of the proportions of pfcrt T76 allele from 2000 to 2009. We experienced first a decrease until 2005 followed by an increase from 2006 to 2009.
The decrease might be explained by an advantage of the pfcrt K76 allele over its mutant form after removal of CQ pressure. The following re-increase of the mutant allele from 2005 is probably caused by a survival of this allele within P. falciparum isolates circulating in the Pikine region. The same trend has been observed in Vietnam where CQ is still prescribed for Plasmodium vivax treatment (Isozumi et al. 2010) and in Dielmo, in a strictly controlled environment free of CQ pressure (Noranate et al. 2007). In the latter village, local populations perpetuated a mutant parasite population that remained essentially without drug pressure. In the Pikine’s context, a contribution of resistant alleles from elsewhere is likely as this area welcomes many people coming from the other regions of Senegal from which we do not have details about the circulation of the mutant alleles.
One should acknowledge that CQ might still circulate within the area exerting CQ pressure because the official discontinuation of CQ use was not followed by a total withdrawal of the molecule from the non-official market as in the Central African Republic (Manirakiza et al. 2010). Indeed in Senegal, it is common for families to purchase very affordable drugs in the non-official market. The origin and the type of these drugs are not fully investigated; however, we consider that CQ might be included, driving sustained presence of mutant pfcrt. In local health centers, ACTs are officially free of charge for malaria-infected people; however, the patients have to afford first consultation fees that some consider as expensive leading them to buy cheaper medicines in the black market. A survey conducted in the southeastern region of Senegal has evidenced that 40 % of children with fever were not taken promptly to hospital to seek treatment because of self medication with drugs already in the home (Smith et al. 2010).
The other factor that might explain the re-increase of the prevalence of pfcrt T76 is that CQ has been replaced as a first-line treatment of uncomplicated malaria by two consecutive combination therapies in which amodiaquine was always present. One can speculate about the possible contribution of amodiaquine, which is chemically and functionally related to CQ, in preventing a further decrease of the prevalence of pfcrt T76 in 2005 followed by a subsequent increase of the proportion of the same mutation.
Although the prevalence of pfcrt T76 has changed greatly over the years, pfmdr1 Y86 mutation that is also associated with CQR (Duraisingh et al. 1997) has shown a modest decrease in frequency from 2000 to 2009 without any statistical significance. The official discontinuation of CQ has had no significant effect on pfmdr1 Y86. This finding suggests the involvement of other factors.
Our molecular analyses show a clear pattern of linkage disequilibrium within this parasite population as described in Guinea Bissau (Ursing et al. 2009), Nigeria, (Adagu and Warhurst 2001; Happi et al. 2006), and Venezuela (Griffing et al. 2010). We consider very likely that this pattern is due to the use of CQ that populations can afford in non-official markets.
Today, both ACTs regimens artesunate–amodiaquine and artemether–lumefantrine are widely used in local health facilities with the latter representing officially the first-line treatment. However in reality, from time to time there are shortages of one ACT, the other one becoming then the available option. The pattern of opposing selective pressure caused by both combinations has been documented elsewhere in Africa with amodiaquine selecting for pfmdr1 Y86 (Dokomajilar et al. 2006; Holmgren et al. 2006) and pfcrt T76 (Holmgren et al. 2007) while lumefantrine selects for pfmdr1 N86 (Sisowath et al. 2005; Some et al. 2010) and pfcrt K76 (Sisowath et al. 2009; Some et al. 2010).
The long half-life of the principal active metabolite of amodiaquine 9–18 days (Pussard et al. 1987) and moderate half-life of lumefantrine 3–6 days (Travassos and Laufer 2009) is an advantage in the Senegalese context where malaria endemicity is mostly low. Indeed malaria parasites can be eliminated before they develop resistance. However the long-term consequence of the selection by both drugs is unknown.
In contrast to the molecular data that were collected routinely in a long-term surveillance strategy, the in vitro CQR results were obtained episodically in 2000, 2001, and 2009. The lack of the monoclonal antibodies used in the DELI test prevented us from conducting the assays during some seasons. However for comparison purposes between data generated in 2000, 2001, and 2009, the same protocols were used and, in addition, the same person performed the assays. Therefore, we expect that our findings can be used as an indicator of the level of CQR as this has not been determined since the official withdrawal of the molecule. Our results highlight significant changes of the in vitro susceptibility to CQ between 2000 and 2009. The geometric mean IC50 that peaked in 2001, decreased dramatically in 2009. We anticipate that the reduction of CQ pressure within the P. falciparum population circulating in Pikine may account for the surge of isolates susceptible to the molecule.
To conclude, despite the official CQ removal, there are still substantial in vitro CQR strains as well as parasites harboring mutant pfcrt and pfmdr1 alleles circulating in Pikine. This finding illustrates that CQ is still widely used within this area. Consequently, more effort should be done by the health authorities to fully remove the molecule from the black market. Additionally, health authorities should monitor closely the two ACTs that are used throughout the country especially because the long-term consequence of selection by the artemisinin derivatives partner drugs is unknown.
References
Adagu IS, Warhurst C (2001) Plasmodium falciparum: linkage disequilibrium between loci in chromosomes 7 and 5 and chloroquine selective pressure in Northern Nigeria. Parasitology 123:219–224
Alker AP, Lim P, Sem R, Shah NK, Yi P, Bouth DM, Tsuyuoka R, Maguire JD, Fandeur T, Ariey F, Wongsrichanalai C, Meshnick SR (2007) Pfmdr1 and in vivo resistance to artesunate mefloquine in falciparum malaria in the Cambodian–Thai border. AmJTrop Med Hyg 76:641–647
Basco LK, Ringwald P (2000) Molecular epidemiology of malaria in Yaounde, Cameroon. VI. Sequence variations in the Plasmodium falciparum dihydrofolate reductase-thymidylate synthase gene and in vitro resistance to pyrimethamine and cycloguanil. AmJTrop Med Hyg 62:271–276
Djimde A, Doumbo OK, Cortese JF, Kayentao K, Doumbo S, Diourte Y, Dicko A, Su XZ, Nomura T, Fidock DA, Wellems TE, Plowe CV (2001) A molecular marker for chloroquine-resistant falciparum malaria. N Engl J Med 344:257–263
Dokomajilar C, Lankoande ZM, Dorsey G, Zongo I, Ouedraogo JB, Rosenthal PJ (2006) Roles of specific Plasmodium falciparum mutations in resistance to amodiaquine and sulfadoxine–pyrimethamine in Burkina Faso. AmJTrop Med Hyg 75:162–165
Dondorp A, Nosten F, Yi P, Das D, Phyo AP, Tarning JT, Lwin KM, Ariey M, Hanpithakpong W, Lee SJ, Ringwald P, Silamut K, Imwong M, Chotivanich K, Lim P, Herdman T, An SS, Yeung S, Singhasivanon P, Day NP, Lindergardh N, Socheat D, White NJ (2009) Artemisinin resistance in Plasmodium falciparum malaria. N Engl J Med 361:455–467
Duraisingh MT, Drakeley CJ, Muller O, Bailey R, Snounou G, Targett GAT, Greenwood BM, Warhurst DC (1997) Evidence for selection for the tyrosine-86 allele of the pfmdr 1 gene of Plasmodium falciparum by chloroquine and amodiaquine. Parasitology 114:205–211
Faye B, Ndiaye JL, Ndiaye D, Dieng Y, Faye O, Gaye O (2007) Efficacy and tolerability of four antimalarial combinations in the treatment of uncomplicated Plasmodium falciparum malaria in Senegal. Malar J 6:80
Fidock DA, Nomura T, Talley AK, Cooper RA, Dzekunov SM, Ferdig MT, Ursos LM, Sidhu AB, Naude B, Deitsch KW, Su XZ, Wootton JC, Roepe PD, Wellems TE (2000) Mutations in the Plasmodium falciparum digestive vacuole transmembrane protein pfCRT and evidence for their role in chloroquine resistance. Mol Cell 6:861–871
Gaye O, Bah IB, Diallo S, Victorius A, Bengua E, Faye O (1990) The emergence of chloroquine-resistant malaria in Dakar, Senegal. Ann Soc Belg Med Trop 70:33–37
Gaye O, Babou I, Faye O, Fall A, Molez JF, Bah IB, Diallo S (1993) Malaria morbidity and therapeutic efficacy of antimalarials. Study conducted in the region of Dakar. Med Trop 53:479–485
Griffing S, Syphard L, Sridaran S, McCollum AM, Mixson-Hayden T, Vinayak S, Villegas L, Barnwell JW, Escalante AA, Udhayakumar V (2010) Pfmdr1 amplification and fixation of pfcrt chloroquine resistance alleles in Plasmodium falciparum in Venezuela. Antimicrob Agents Chemother 54:1572–1579
Happi CT, Gbotosho GO, Folarin OA, Sowunmi A, Bolaji OM, Fateye BA, Kyle DE, Milhous W, Wirth DF, Oduola AM (2006) Linkage disequilibrium between two distinct loci in chromosome 5 and 7 of Plasmodium falciparum and in vivo chloroquine resistance in Southwest Nigeria. Parasitol Res 100:141–148
Holmgren G, Gil JP, Ferreira PM, Veiga MI, Obonyo CO, Bjorkman A (2006) Amodiaquine resistant Plasmodium falciparum malaria in vivo is associated with selection of pfcrt 76T and pfmdr1 86Y. Infect Genet Evol 6:309–314
Holmgren G, Hamrin J, Svard J, Martensson A, Gil JP, Bjorkman A (2007) Selection of pfmdr1 mutations after amodiaquine monotherapy and amodiaquine plus artemisinin therapy in East Africa. Infect Genet Evol 7:562–569
Isozumi R, Uemura H, Le DD, Truong VH, Nguyen DG, Ha VV, Bui QP, Nguyen VT, Nakazawa S (2010) Longitudinal survey of Plasmodium falciparum infection in Vietnam: characteristics of antimalarial resistance and their associated factors. J Clin Microbiol 48:70–77
Krogstad DJ, Gluzman IY, Kyle DE, Oduala AM, Martin SK, Milhous WK, Schlesinger PH (1987) Efflux of chloroquine from Plasmodium falciparum: mechanism of chloroquine resistance. Science 238:1283–1285
Kublin JG, Cortese JF, Njunju EM, Mukadam AG, Wirima JJ, Kazembe PN, Djimde AA, Kouriba B, Taylor TE, Plowe CV (2003) Reemergence of chloroquine-sensitive Plasmodium falciparum malaria after cessation of chloroquine use in Malawi. J Infect Dis 187:1870–1875
Laufer MK, Thesing PC, Eddington ND, Masonga R, Dzinjalamala FK, Takala SL, Taylor TE, Plowe CV (2006) Return of chloroquine antimalarial efficacy in Malawi. N Engl J Med 355:1959–1966
Manirakiza A, Njuimo SP, Le Faou A, Malvy D, Millet P (2010) Availability of antimalarial drugs and evaluation of the attitude and practices for the treatment of uncomplicated malaria in Bangui, Central African Republic. J Trop Med 2010:510834
Mita T, Kaneko A, Lum KJ, Bwijo B, Takechi M, Zungu IL, Tsukahara T, Tanabe K, Kobayakawa T, Bjorkman A (2003) Recovery of chloroquine sensitivity and low prevalence of the Plasmodium falciparum chloroquine resistance transporter gene mutation K76T following the discontinuance of chloroquine use in Malawi. AmJTrop Med Hyg 68:413–415
Moreno A, Brasseur P, Cuzin-Ouattara N, Blanc C, Druilhe P (2001) Evaluation under field conditions of the colorimetric DELI-microtest for the assessment of Plasmodium falciparum drug resistance. Trans R Soc Trop Med Hyg 95:100–103
Mwai L, Ochong E, Abdirahman A, Kiara SM, Ward S, Kokwaro G, Sasi P, March K, Borrman S, Mackinnon M, Nzila A (2009) Chloroquine resistance before and after its withdrawal in Kenya. Malar J 8:106
Ndiaye D, Patel V, Demas A, LeRoux M, Mboup S, Ndir O, Clardy J, Lakshmanan V, Daily JP, Wirth DF (2010) A non-radioactive DAPI-based high throughput in vitro assay to assess Plasmodium falciparum responsiveness to antimalarials increased sensitivity of P. falciparum to chloroquine in Senegal. AmJTrop Med Hyg 82:228–230
Noedl H, Socheat D, Satimai W (2009) Artemisinin-resistant malaria in Asia. N Engl J Med 36:540–541
Noranate R, Durand R, Tall A, Marama L, Spiegel A, Sokhna C, Pradines B, Cojean S, Guillotte M, Bischoff E, Ekala MT, Bouchier C, Fandeur T, Ariey F, Patarapotikul J, Lebras J, Trape JF, Rogier C, Mercereau-Puijalon O (2007) Rapid dissemination of Plasmodium falciparum drug resistance despite strictly controlled antimalarial use. PLoS One 2(1):e139
Pussard E, Verdier F, Faurisson F, Scherrmann JM, Le Bras J, Blayo MC (1987) Disposition of monodesethylamodiaquine after a single oral dose of amodiaquine and three regimens forprophylaxis against Plasmodium falciparum malaria. Eur J Clin Pharmacol 33:409–414
Reed MB, Saliba KJ, Caruana SR, Kirk K, Cowman AF (2000) Pgh1 modulates sensitivity and resistance to multiple antimalarials in Plasmodium falciparum. Nature 403:906–909
Sarr O, Myrick A, Daily JP, Diop BM, Dieng T, Ndir O, Sow PS, Mboup S, Wirth DF (2005) In vivo and in vitro analysis of chloroquine resistance in Plasmodium falciparum isolates from Senegal. Parasitol Res 97:136–140
Sarr O, Ahouidi A, Daily J, Ly O, Ndiaye D, Dieng T, Ndir O, Mboup S, Wirth DF (2008) Mutations in pfcrt K76T do not correlate with sulfadoxine pyrimethamine amodiaquine failure in Pikine, Senegal. Parasitol Res 103:765–769
Schwenke A, Brandts C, Philipps J, Winkler S, Werndorfer WH, Kremsner PG (2001) Declining chloroquine resistance of Plasmodium falciparum in Lambarene, Gabon from 1992 to 1998. Wien Klin Wochenschr 113:63–64
Sisowath C, Stromberg J, Martensson A, Msellem M, Obondo C, Bjorkman A, Gil JP (2005) In vivo selection of Plasmodium falciparum pfmdr1 86N coding alleles by artemether–lumefantrine (Coartem). J Infect Dis 191:1014–1017
Sisowath C, Petersen I, Veiga MI, Mårtensson A, Premji Z, Björkman A, Fidock DA, Gil JP (2009) In vivo selection of Plasmodium falciparum parasites carrying the chloroquine-susceptible pfcrt K76 allele after treatment with artemether–lumefantrine in Africa resistance to lumefantrine might be caused by the extended elimination half-life of the molecule. J Infect Dis 199:750–757
Smith LA, Bruce J, Gueye L, Helou A, Diallo R, Gueye B, Jones C, Webster J (2010) From fever to anti-malarials: the treatment seeking-process in rural Senegal. Malar J 9:333
Sokhna CS, Molez JF, Ndiaye P, Sane B, Trape JF (1997) In vivo chemosensitivity tests of Plasmodium falciparum to chloroquine in Senegal: the development of resistance and the assessment of therapeutic efficacy. Bull Soc Pathol Exot 90:83–89
Some AF, Sere YY, Dokomajilar C, Zongo I, Rouamba N, Greenhouse B, Ouedraogo JB, Rosenthal PJ (2010) Selection of known Plasmodium falciparum resistance-mediating polymorphisms by artemether–lumefantrine and amodiaquine–sulfadoxine–pyrimethamine but not dihydroartemisinin–piperaquine in Burkina Faso. Antimicrob Agents Chemother 54:1949–1954
Thomas MT, Ndir O, Dieng T, Mboup S, Wypij D, Maguire JH, Wirth DF (2002) In vitro chloroquine susceptibility and PCR analysis of Pfcrt and Pfmdr1 polymorphisms in Plasmodium falciparum isolates from Senegal. AmJTrop Med Hyg 66:474–480
Trape JF, Legros F, Ndiaye P, Konate L, Bah IB, Diallo S, Verdier F, Hatin I, Le Bras J (1989) Chloroquine-resistant Plasmodium falciparum malaria in Senegal. Trans R Soc Trop Med Hyg 83:761
Travassos MA, Laufer MK (2009) Resistance to antimalarial drugs: molecular, pharmacological and clinical considerations. Pediatr Res 65:64(R)–70(R)
Ursing J, Kofoed PE, Rodrigues A, Rombo L (2009) No seasonal accumulation of resistant P. falciparum when high-dose chloroquine is used. PLoS One 4(8):e6866
Wang X, Mu J, Li G, Chen P, Guo X, Fu L, Chen L, Su X, Wellems TW (2005) Decreased prevalence of the Plasmodium falciparum chloroquine resistance transporter 76T marker associated with cessation of chloroquine use against P. falciparum malaria in Hainan, People’s Republic of China. AmJTrop Med Hyg 72:410–414
World Health Organization (2010). World Malaria Report 2010 116–118. http://www.who.int/malaria/world_malaria_report_2010/en/index.html. Accessed 1 June 2012
Acknowledgments
A NIH Grant to Prof DF Wirth (1.2D43TW01503) and a fellowship from Iwerliewen Fir Bedreete Volleker to Mr. Papa Elhadji Omar Gueye funded the study. We are thankful to patients at the outpatient clinic in Pikine who voluntarily consented to participate in the study.
Conflict of interest
The authors declare that they have no competing financial and personal interests that may influence their work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ly, O., Gueye, P.E.O., Deme, A.B. et al. Evolution of the pfcrt T76 and pfmdr1 Y86 markers and chloroquine susceptibility 8 years after cessation of chloroquine use in Pikine, Senegal. Parasitol Res 111, 1541–1546 (2012). https://doi.org/10.1007/s00436-012-2994-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-012-2994-7